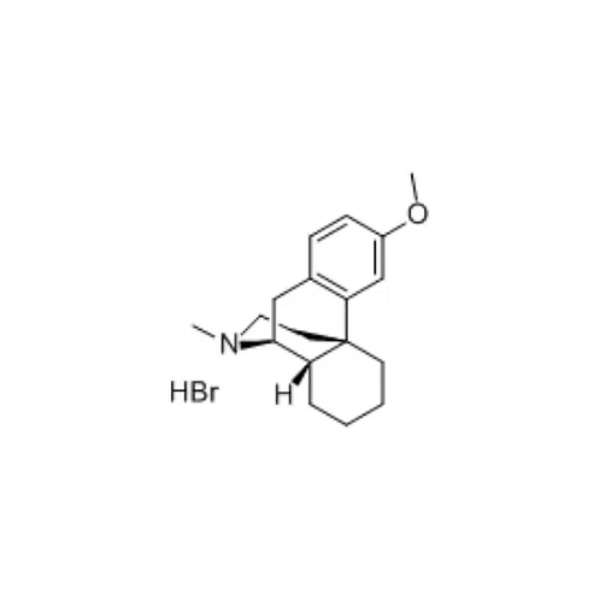
Biperiden Hydrochloride
Biperiden Hydrochloride Specification
- Solubility
- Soluble in water, ethanol, and chloroform
- Smell
- Odorless
- Particle Size
- Micronized powder
- Heavy Metal (%)
- 0.001%
- Storage
- Store in a cool, dry place, protected from light
- Poisonous
- Yes (toxic if ingested in large quantities)
- Ph Level
- 5.5 - 7.5 (1% aqueous solution)
- HS Code
- 29420090
- Structural Formula
- C21H29NOHCl (as per structure)
- Boiling point
- Not applicable (decomposes)
- Taste
- Bitter
- Melting Point
- 212-214C
- Loss on Drying
- 0.5%
- Shelf Life
- 24 months
- EINECS No
- 208-157-6
- Molecular Weight
- 347.92 g/mol
- Color
- White to off-white
- Molecular Formula
- C21H29NO.HCl
- Medicine Name
- Biperiden Hydrochloride
- Chemical Name
- Biperiden Hydrochloride
- CAS No
- 514-65-8
- Type
- Pharmaceutical Raw Material
- Grade
- Pharma Grade
- Usage
- Used as an antiparkinsonian agent, effective in the treatment of Parkinsons disease and drug-induced extrapyramidal symptoms
- Purity(%)
- 99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Related Substances
- 0.2%
- Optical Rotation
- +15 to +18 (20C, in water)
- pKa Value
- 9.4
- Packaging
- HDPE container or double polyethylene bags in cardboard drums
- Chloride Content
- Within pharmacopeial limits
- Residual Solvents
- Complies with ICH Q3C
- Endotoxin Level
- NMT 0.25 EU/mg
- Bulk Density
- Approx. 0.4-0.5 g/cm3
- Microbial Limits
- Complies with pharmacopeial standards
- Identification Method
- IR, HPLC, chemical tests
Biperiden Hydrochloride Trade Information
- Minimum Order Quantity
- 50 Kilograms
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 200 Kilograms Per Month
- Delivery Time
- 1 Week
- Sample Policy
- Contact us for information regarding our sample policy
- Main Export Market(s)
- Asia
- Main Domestic Market
- All India
About Biperiden Hydrochloride
Akineton (biperiden hydrochloride) Tablet is an anticholinergic antiparkinson agent used to treat the stiffness, tremors, spasms, and poor muscle control of Parkinsons disease.Sihauli Chemicals is one of the leading Akineton (biperiden hydrochloride) Manufacturer and Exporter form India. Sihauli Chemicals Supply Akineton (biperiden hydrochloride) to Asia, Africa, Europe, North & South America and Oceania.
Our Global Presense:
Asia: Bangladesh, Burma (Myanmar), China, Hong Kong (UK), Indonesia, Iran, Israel, Japan, Jordan, Kampuchea (Cambodia), Kuwait, Laos, Lebanon, Macao (Portuguese), Malaysia, Maldives, Mongolia, Nepal, Oman, Philippines, Qatar, Saudi Arabia, Singapore, South Korea, Sri Lanka, Taiwan, Thailand, Turkey, United Arab Emirates, Vietnam.
Europe: Albania, Andorra, Austria, Belgium, Bulgaria, Cyprus, Czechoslovakia, Denmark, Finland, France, German Democratic Republic, Gibraltar, Greece, Hungary, Iceland, Ireland, Italy, Liechtenstein, Luxembourg, Malta, Monaco, Netherlands, Norway, Poland, Portugal, Romania, San Marino, Spain, Sweden, Switzerland, United Kingdom, U.S.S.R., Yugoslavia. Africa: Algeria, Angola, Egypt, Ethiopia, Gabon, Ghana, Kenya, Liberia, Libya, Madagascar, Mauritius, Morocco, Mozambique, Namibia, Nigeria, Reunion (France), Seychelles, Somalia, South Africa, Sudan, Tanzania, Tunisia, Uganda, Zambia, Zimbabwe.
North America: Bahamas, Barbados, Bermuda, British Virgin Islands, Canada, Costa Rica, Cuba, Dominica, El Salvador, Greenland (Denmark), Grenada, Guatemala, Haiti, Honduras, Jamaica, Martinique, Mexico, Montserrat, Nicaragua, Panama, Puerto Rico (USA), St. Vincent & the Grenadines, Trinidad & Tobago, USA, Virgin Islands.
South America: Argentina, Bolivia, Brazil, Chile, Colombia, Ecuador, French Guiana, Guyana, Paraguay, Peru, Venezuela.
Oceania: Australia, New Zealand, Solomon Islands, Wallis & Futuna Islands (France).
Key Advantages of Biperiden Hydrochloride
Biperiden Hydrochloride is renowned for its efficacy in managing symptoms of Parkinsons disease and extrapyramidal disorders induced by other medications. Its high purity and strict compliance with international standards ensure safety and consistent therapeutic performance. Its stability, solubility in several solvents, and convenient solid form make it suitable for various pharmaceutical formulations. Careful packaging further preserves its quality during storage and transportation.
Quality and Safety Compliance
Manufactured under stringent quality controls, Biperiden Hydrochloride aligns with major pharmacopeial standards. With related substances limited to less than 0.2% and heavy metals at no more than 0.001%, it upholds exceptional chemical purity. Testing via IR, HPLC, and chemical methods validates its identity. Shelf life of 24 months and appropriate storage recommendations ensure long-term stability and reduced risk of contamination.
FAQs of Biperiden Hydrochloride:
Q: How is the identification of Biperiden Hydrochloride performed?
A: Biperiden Hydrochloride is identified using infrared (IR) spectroscopy, high-performance liquid chromatography (HPLC), and specific chemical tests. These methods ensure the authenticity and purity of the substance adheres to pharmacopeial standards.Q: What is the recommended storage condition for Biperiden Hydrochloride?
A: The product should be stored in a cool, dry place, protected from light, and kept in tightly closed HDPE containers or double polyethylene bags within cardboard drums to maintain stability and prolong shelf life.Q: When does the shelf life of Biperiden Hydrochloride expire?
A: The shelf life of Biperiden Hydrochloride is 24 months from the date of manufacture when stored under recommended conditions, after which its quality and efficacy may decline.Q: Where is Biperiden Hydrochloride manufactured and supplied?
A: Biperiden Hydrochloride is primarily manufactured, exported, and supplied by pharmaceutical companies in India, with global distribution to meet varied clinical needs.Q: What are the main uses and benefits of Biperiden Hydrochloride?
A: This compound is mainly used as an antiparkinsonian agent, providing relief from Parkinsons disease symptoms and mitigating drug-induced extrapyramidal effects. Its high purity and compliance with quality standards support safe therapeutic use.Q: How is the bulk density and physical form relevant to pharmaceutical processes?
A: Biperiden Hydrochloride is supplied as a micronized, free-flowing crystalline powder with a bulk density of approximately 0.40.5 g/cm. This property aids in precise dosing and uniform mixing during pharmaceutical manufacturing.Q: What measures are in place to ensure the safety of Biperiden Hydrochloride?
A: Safety is ensured by strict control over residual solvents, microbial limits, heavy metals, and endotoxin content, all within pharmacopeial and ICH Q3C guidelines. This minimizes contamination risks and supports product safety for end-users.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Chemicals Category
Dextromethorphan Hydrobromide
Price 5350.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Purity(%) : 99%
CAS No : 125699
Physical Form : Powder
HS Code : 29333990
5949-29-1 Citric Acid Monohydrate
Price 799.0 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Purity(%) : 99%
CAS No : 5949291
Physical Form : Powder
120-80-9 98 Percent Catechol Chemical
Price 2100 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Purity(%) : 98%
CAS No : 120809
Physical Form : Powder
Lanoconazole RAW MATERIAL
Price 5001.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Purity(%) : 98 99%
CAS No : 101530103
Physical Form : Powder
HS Code : 29349990