Lead Monoxide (Litharge, Massicot, Lead oxide)
Lead Monoxide (Litharge, Massicot, Lead oxide) Specification
- Ph Level
- Amphoteric in nature
- Size
- Fine Powder
- Flash Point
- Non-flammable
- Melting Point
- 888C
- Packaging Type
- 25 kg and 50 kg HDPE bags/Drums
- Density
- 9.53 Gram per cubic centimeter(g/cm3)
- Purity
- 99%
- Shelf Life
- 24 months
- Physical State
- Powder
- Storage Instructions
- Store in tightly closed container in a cool, dry, and well-ventilated area
- Boiling point
- 1472C
- Molecular Weight
- 223.2 g/mol
- Molecular Formula
- PbO
- CAS No
- 1317-36-8
- Usage
- Used as raw material for lead glass, frit glazing, vulcanization of rubber, lead-acid battery plates, pigments
- Grade
- Industrial Grade
- Type
- Inorganic Compound
- Application
- Glass, ceramics, paints, batteries, rubber, lubricants, and chemical synthesis
- Appearance
- Yellow or red powder
- Purity(%)
- 99%
- EINECS No
- 215-267-0
- Appearance Variation
- Yellow litharge (-PbO), Red massicot (-PbO), color depends on crystal structure
- Lead Content
- Minimum 93%
- Particle Size
- 1-10 microns
- Synonyms
- Litharge, Massicot, Lead oxide
- Refractive Index
- 2.6 - 2.8
- Stability
- Stable under normal conditions
- UN Number
- UN 2291
- Moisture Content
- <0.5%
- Storage Compatibility
- Keep away from foodstuffs, acids, and oxidizers
- Minimum Order Quantity
- 500 kg
- Hazard Class
- 6.1 (Toxic Substance)
- Odor
- Odorless
- Solubility
- Insoluble in water, soluble in acids and alkalis
Lead Monoxide (Litharge, Massicot, Lead oxide) Trade Information
- Minimum Order Quantity
- 50 Kilograms
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 200 Kilograms Per Month
- Delivery Time
- 1 Week
- Sample Policy
- Contact us for information regarding our sample policy
- Main Export Market(s)
- Asia
- Main Domestic Market
- All India
About Lead Monoxide (Litharge, Massicot, Lead oxide)
Lead Monoxide, also known as litharge (-PbO) or massicot (I2-PbO), is an inorganic compound with significant industrial applications. It appears in two polymorphs: a red or yellow tetragonal form known as litharge and a yellow orthorhombic form known as massicot.Versatile Industrial Applications
Lead Monoxide serves as a foundational material in industries such as glass manufacturing, ceramics, pigment production, batteries, and rubber vulcanization. Its high purity and consistent particle size make it essential in producing lead-acid battery plates, frit glazing, and specialty glass. The compound is valued for enhancing material properties like durability, color stability, and chemical reactivity.
Physical and Chemical Stability
PbO is stable under normal conditions and non-flammable, with a melting point of 888C and a boiling point of 1472C. It features a high density of 9.53 g/cm3, and its moisture content is kept below 0.5%. The product's amphoteric nature allows it to react with both acids and alkalis, making it highly adaptable in chemical industries.
Safe Storage, Handling, and Packaging
As a toxic substance (Hazard Class 6.1), Lead Monoxide requires careful handling. Store it in tightly sealed containers, away from foodstuffs, acids, and oxidizers, in a cool, dry, and well-ventilated area. Standard packaging includes 25 kg and 50 kg HDPE bags or drums, ensuring safe transport and storage, with a shelf life of up to 24 months.
FAQ's of Lead Monoxide (Litharge, Massicot, Lead oxide):
Q: How should Lead Monoxide (Litharge, Massicot) be stored to ensure safety?
A: Lead Monoxide must be stored in tightly closed containers, away from foodstuffs, acids, and oxidizing agents. It should be placed in a cool, dry, and well-ventilated area to prevent contamination and preserve its quality for up to 24 months.Q: What are the key applications of Lead Monoxide in industry?
A: Lead Monoxide is primarily used in glass manufacturing, ceramics, paints, batteries (notably lead-acid battery plates), rubber vulcanization, lubricants, pigments, and as a reagent in chemical synthesis. Its high purity and versatile reactivity make it valuable in these fields.Q: When is Lead Monoxide typically required to be handled with additional safety precautions?
A: Due to its toxicity (Hazard Class 6.1, UN 2291), strict safety measures must be practiced during storage, handling, and transport. This includes using protective equipment and following regulated procedures to avoid exposure and environmental contamination.Q: Where is Lead Monoxide commonly produced and supplied from?
A: In India, Lead Monoxide is manufactured, supplied, exported, and imported by various industrial enterprises specializing in inorganic compounds, with minimum order quantities often starting at 500 kg.Q: What benefits does Lead Monoxide offer in glass and ceramic production?
A: Lead Monoxide improves the density, refractive index, and color stability of glass and ceramics, resulting in enhanced brightness and resilience. Its amphoteric nature facilitates interaction with other compounds during synthesis.Q: How does the crystal structure of Lead Monoxide influence its appearance and properties?
A: PbO occurs as yellow litharge (-PbO) or red massicot (-PbO), with color determined by the crystal form. Both forms exhibit similar industrial uses, but crystal structure can affect the material's optical properties.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Laboratory Chemicals Category
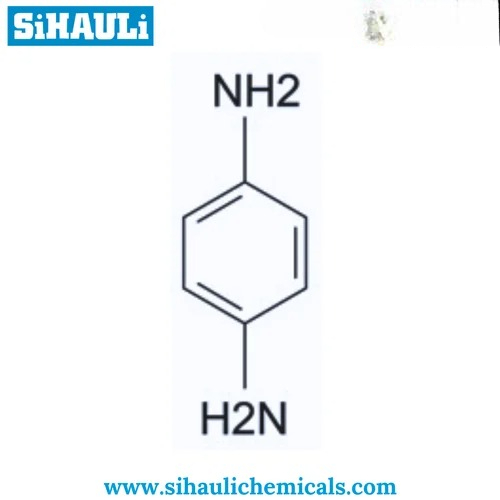
Para Phenylenediamine chemicals Manufacturers, Suppliers, Exporters From Vasai Mumbai India
Price 200 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Physical State : Solid (Crystalline)
Purity : 99% Min
Shelf Life : 24 Months
Molecular Formula : C6H8N2
Poly Ethanolamine Chemical Manufacturers, Suppliers, Exporters From Vasai
Price 110 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Physical State : Liquid
Purity : 99%
Shelf Life : 24 months
Molecular Formula : C2H7NO
Tetrabutylammonium bromide (TBAB) Manufacturers, Suppliers, Exporters From Vasai Mumbai India
Price 480 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Physical State : Solid
Purity : >=99%
Shelf Life : 2 years
Molecular Formula : C16H36BrN
Acetone (Dimethyl ketone)
Price 5347.0 INR / Liter
Minimum Order Quantity : 25 Liters
Physical State : Liquid
Purity : 99.5% min
Shelf Life : 24 months in sealed container
Molecular Formula : C3H6O